Thanks to Monty at Centurion Arms for his help with the recessed barrel crown and flush cut slide stop pin. 
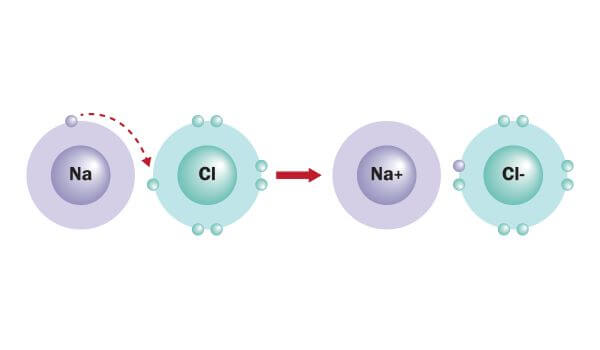
Other parts include an EGW HD Hammer, Sear and Disconnector, 10-8 flat trigger, 10-8 rear sight, 10-8 fiber optic front, 10-8 mag catch, 10-8 grip screws, 10-8 slide stop, 10-8 recoil spring plug, an aggressively recontoured Wilson Combat ambi-safety, CMC grip safety, and STI hammer strut. It works great with the 10-8 Performance base pads. The front is relieved so that stuck magazines can be yanked out by the toe of the magazine. Springfield Custom did a beautiful job with the machine checkering of the front strap and welding of the Legacy Custom magazine well, which adds no net length to the frame yet nearly doubles the margin for error for a magazine change. I managed to get a hold of a rare IMI Match barrel which I thought would be perfect for this project. While metals share electrons in the third orbital, transition metals share third and fourth orbital electrons.A couple years ago, I started building on a Colt Series 70 Reproduction I had purchased through the Colt Armorer’s Program nearly 10 years prior. These types of elements have even higher melting and boiling temperatures than metals do because they share more valence electrons. Some elements are called transitional metals. The same thing happens when magnesium or other metals metallically bond to themselves. For example, when sodium metallically bonds with itself, each atom is sharing the electrons in the third orbital with up to eight other atoms. Metallic bonds are defined as those in which metals share valence electrons. Most elements are metals, including some such as sodium, radium and calcium, which may not seem very metallic. While many elements are commonly known as metals, including iron, aluminum, gold, silver and nickel, metals include a variety of other elements as well. Metals are the only substances that use metallic bonds among their atoms. The Composition of Metals in Metallic Bonding The result is an orderly structure of positive metal atoms surrounded by a sea of negative electrons that hold the ions together like glue. Once the valence electrons detach from their original atomic owners and float around in the sea, the metal atoms become positive ions. The golf balls are the metal atoms, and the water represents the valence electrons shared by all of the atoms. What you now have is something like metallic bonding. Do you see any spaces between the balls? If you turn on the faucet and plug the drain, the water will fill up those spaces. The golf balls will arrange themselves in an orderly fashion as they fill the space in the tub. Imagine filling your bathtub with golf balls. The metallic bond is not the easiest type of bond to understand, so an analogy might help. The big pool is like a free-for-all in that any valence electron can move to any atom within the material. 
All of the atoms in that small piece of metal are sharing a big pool of valence electrons known as a sea of electrons or delocalized electrons. Take a look at your desk and see if you can find a small piece of metal like a paper clip or a staple. Instead of a bond between just two atoms, a metallic bonds is a sharing of electrons between many atoms of a metal element. How Metallic Bonding Works?Ī metallic bond is pretty different from covalent and ionic bonds, but the goal is the same: to achieve a lower energy state. When one atom takes an electron away from another and the resulting positive and negative ions are attracted to each other, those atoms have formed an ionic bond. When two atoms share electrons, they form a covalent bond. If you have already learned about covalent and ionic bonding in metallic bonds, you know that these bonds occur between two atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
